Value Creation with Safety & Security Sector
Providing Solutions Contributing Security and Safety in Response to Various Lifestyles



Corresponding SDGs
The JVCKENWOOD Group has identified the material issue of "Safety & Security Initiatives" to achieve the future it envisions by 2030. In the Safety & Security Sector, we contribute to safety and security with products such as professional radio systems, emergency and professional-use broadcasting equipment, and medical monitors.
Contributing to the Creating Safe and Secure Communities
In the Safety & Security Sector, the Company has developed a number of products tailored to each industry and application for the sake of creating safe and secure communities, and we engage in a total solution business utilizing a wide range of distribution channels.
Communications Systems Business
The Communications Systems Business, which originated from the manufacture and sale of amateur radio equipment, has evolved primarily around professional radio communications, leveraging the communication and manufacturing technologies cultivated since our founding. We will continue developing products for the amateur radio market and plan to launch a next-generation in-vehicle amateur radio within fiscal 2025. Our commitment to user-oriented technology has been passed down to our professional radio systems, ensuring the business drives the entire Group.
Amid rising interest globally for crisis management measures, such as for disasters, we have seen a renewed awareness of the importance of independent wireless networks that do not rely on public cellphone networks as a reliable method of communication in mountainous regions and areas experiencing or affected by disasters. They are an essential social infrastructure when creating safe and secure communities as they are highly robust and can be used with peace of mind in a harsh environment. As the shift from analog to digital technology advances, opportunities for creating new value in the broadband domain, which can transmit a high volume of data at fast speeds, have been expanding, and the North American market in particular is expected to continue showing steady demand. Against this backdrop, we aim to achieve revenue of \120.0 billion in fiscal 2030 in the Communications Systems Business, centered on the North American public safety and private sector markets. To realize this, we will aim for sustainable growth under the following two approaches.
Stable and Continuous Provision of Highly Reliable Communications Systems
The Communications Systems Business has a proven track record and customer base that have been built up over 40 years in the public safety market, which requires confidentiality and robustness in communications. Since 2023, our market share in North America has been expanding through the introduction of the VP8000 portable radio and the VM8000 mobile radio, both supporting tri-band*1 operation. In the commercial market, products utilizing our proprietary communication system Trunking 2.5, based on the Digital Mobile Radio standard, have earned high trust. We aim to further expand our market share in both markets by ensuring a stable supply of these products.
*1 Covers three band frequencies: VHF, UHF, and 700/800 MHz
Creation of New Value in the Broadband Domain
The broadband domain is expected to show medium- to long-term growth and its needs have diversified. In the Communications Systems Business, we aim to provide new communications solutions that meet diversified needs, utilizing the strengths cultivated in the narrowband business and strengthening our wireless internet protocol (IP) technological capabilities through cooperation with other companies.
The Company is recognized worldwide for its high quality and reliability, underpinned by our expertise and knowhow accumulated over the years, and for its flexible system proposals that meet customer needs and budgets. As part of our contribution to the creation of safe and secure communities, we have established themes and KPIs as follows, and we are promoting initiatives while managing progress to achieve our goals.
| Theme | Plan/Achievement | KPI | |||
|---|---|---|---|---|---|
| FY2023 | FY2024 | FY2023-FY2025 (Cumulative) |
FY2023-FY2030 (Cumulative) |
||
| Provide a stable means of communication even at the site of a disaster or accident or in an emergency | Plan | Number of professional radio system units sold | |||
| 1.2 | 1.3 | 4 | 12 | ||
| Achievement | 1.3 | 1.2 | |||
| Despite an increase in revenue due to increased demand for high-end radios, revenue has also been impacted by a decrease in the sale of low-end radios. | |||||

Professional Radio System
Please see the links provided below for further details.
Emergency & Professional-Use Multilingual Broadcasting Equipment
JVCKENWOOD makes use of its know-how and technological expertise in acoustics systems to provide broadcasting equipment and evacuation guidance support systems for use in emergencies and for business as part of its lines of products and services designed to prevent and mitigate disasters. We offer a line of broadcasting systems for large-scale office buildings, shopping malls, and factories that play a vital role in regional disaster management. We are advancing the implementation of multilingual emergency and professional-use broadcasting equipment to help everyone in Japan, including non-Japanese nationals, live safely with peace of mind. In fiscal 2019, working under our plan to launch new disaster-prevention and disaster-mitigation products and services, we set a target of installing 50 units of such equipment in response to the growing number of inbound tourists to Japan, and installed 57 units by the end of the fiscal year.
Our emergency and professional-use broadcasting equipment—the EM-1500 series and the EM-K150 series—is able to broadcast voice warning messages in Japanese and English. Furthermore, by replacing the default card with a multilingual CF (compact flash) card, the equipment is able to make announcements in three to four different languages (Chinese and Korean in addition to Japanese and English)*, thus making it possible to reach even more people in times of emergency. Our evacuation guidance support system can be connected to signage, etc., to share messages in the form of video, sound and radio to be delivered to evacuees, on top of the conventional emergency and professional-use broadcasting.
* Approval from the fire department of the local jurisdiction is needed for implementing this equipment.

Multilingual emergency and professional-use broadcasting equipment—rack type
EM-1500 Series

Multilingual emergency and professional-use broadcasting equipment—wall mount type
EM-K150 Series
Please see the links provided below for further details.
Car Number Plate Recognition System
The Car Number Plate Recognition System recognizes the number plate on a vehicle captured on a camera image and automatically checks vehicles entering logistics facilities and commercial facilities. The system enables entry and exit log control and contributes to reducing the workload of receptionists and guards. Furthermore, the system can be used to identify the number of entries into and exits from a facility on a real-time basis or automatically announce the entry and exit of a vehicle that requires attention if the car plate number is registered in advance, thereby assisting in managing the security and safety of facilities.
In addition, in response to the '2024 logistics problem' - in which legal restrictions on driver overtime may cause a shortage of drivers - we have linked the site entry and exit records to the berth reservation system, providing visibility of the waiting time and cargo handling time as well as saving labor in entry and exit management.
For details, please see the link provided below.
Video analysis solution: “Car Number Plate Recognition System” (Japanese)
Healthcare Initiatives
The JVCKENWOOD Group develops products for the healthcare industry by making use of the unique expertise that we possess in image and video processing technologies. Through the development of high-precision medical image display monitors, we are supporting the front lines of the medical field to deliver appropriate medical services to as many people as possible. The healthcare business is also a field that will have a strong impact on the super-aging society, and, as such, we are striving to develop and launch products that meet the needs of medical professionals.
32 inch High-Resolution Display of 8 Million Pixels
In recent years, various medical imaging data such as CT and MRI have been handled on the front lines of medical image diagnosis due to the evolution and diversification of medical equipment. In response, the Company provides high-precision medical image display monitors.
The CL-R813 is the largest medical image display monitor manufactured by JVCKENWOOD, with a 32 inch wide screen area and a high-resolution display of 8 million pixels. This monitor can simultaneously display six high-resolution CT or X-ray fluoroscopy images on a single screen, as well as freely lay out multiple application screens, including viewers, reports, and AI judgment results. As a result, eye movement when reading images is reduced, contributing to improved work efficiency and reduced burden on doctors.
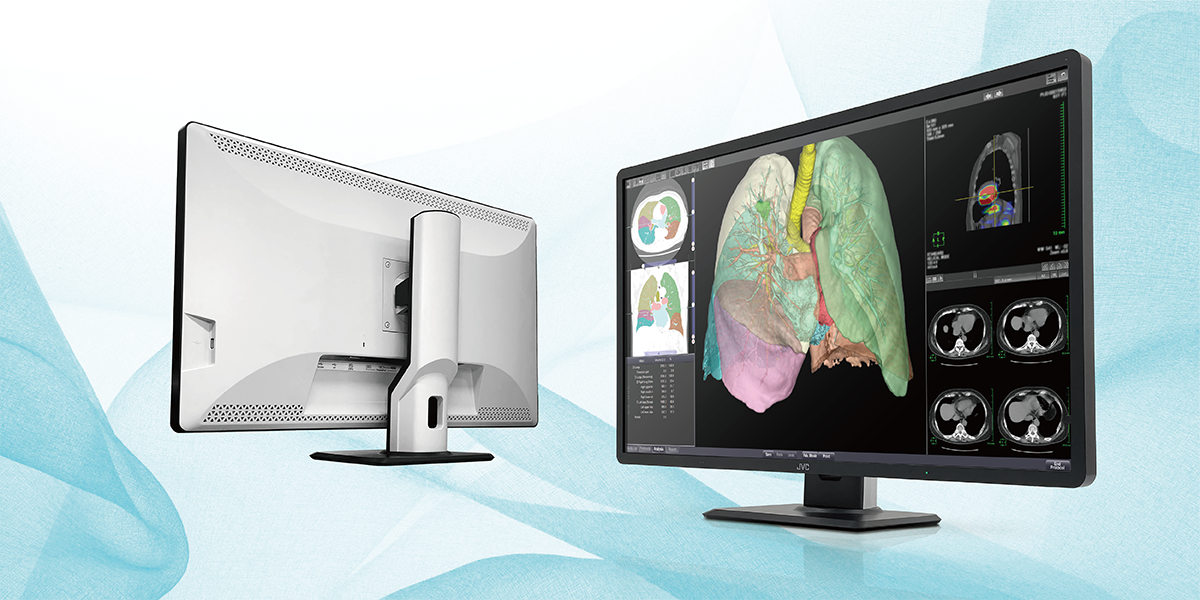
Medical Monitor 「CL-R813」
For details, please see the link provided below.
21.3 inch 3 Million Pixel Color LCD Monitor
In recent years, to address issues such as labor shortages, long working hours, and diversification of work styles in the field of medical diagnostic imaging, initiatives to reform work styles are required, such as improving the working environment, increasing work efficiency, and introducing reading of images from home and satellite image reading rooms. At the same time, the introduction of home image reading has not progressed due to challenges such as the required capital investment and the establishment of a reading environment at home that is equivalent to that of a medical facility. To address this, the JVCKENWOOD has launched the CL-S301, which supports remote and home image reading, for which demand has been growing in recent years.
The CL-S301 is our company's first 21.3 inch 3 million pixel color LCD monitor that supports USB Type-C (Picture Archiving and Communication System) for the PACS medical image management system. This monitor thus enables video transmission and power supply with a single USB Type-C cable, allowing smooth connectivity to notebook PCs and mobile devices. This eliminates the need to carry the previously required AC adapter and multiple cables and keeps cables organized to provide a comfortable workspace. In addition, it features a KVM switch allows switching between multiple workstations with a single keyboard and mouse, thus reducing equipment costs and making more efficient use of space. Furthermore, the usability of "turbo luminance" and "visual point mode," our original image reading support functions that are available on our high-end medical image display monitor models, has been enhanced to support greater work efficiency and work style reform.
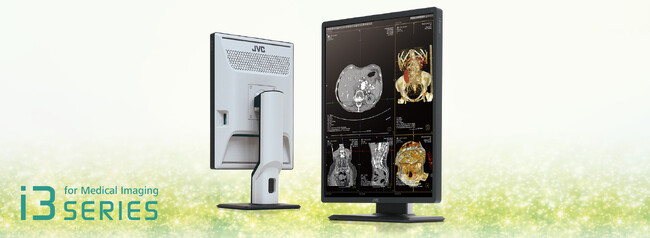
Medical Monitor「CL-S301」
For details, please see the link provided below.

